
Regeneron Just Cured a Type of Deafness With a Single Surgery
Regeneron just landed its first-ever gene therapy approval, and it does something remarkable: restore hearing in children born deaf from a genetic mutation. One surgery, no cochlear implant required. Here's why it matters for a company that built its empire on antibodies.
Imagine being born into silence. Not the peaceful, meditative kind, but a world where you've never heard your mother's voice, a dog bark, or a song on the radio. For roughly 50 babies born each year in the U.S. with a specific genetic mutation, that was the only reality available.
Until last Friday.
A First for a $100 Billion Company
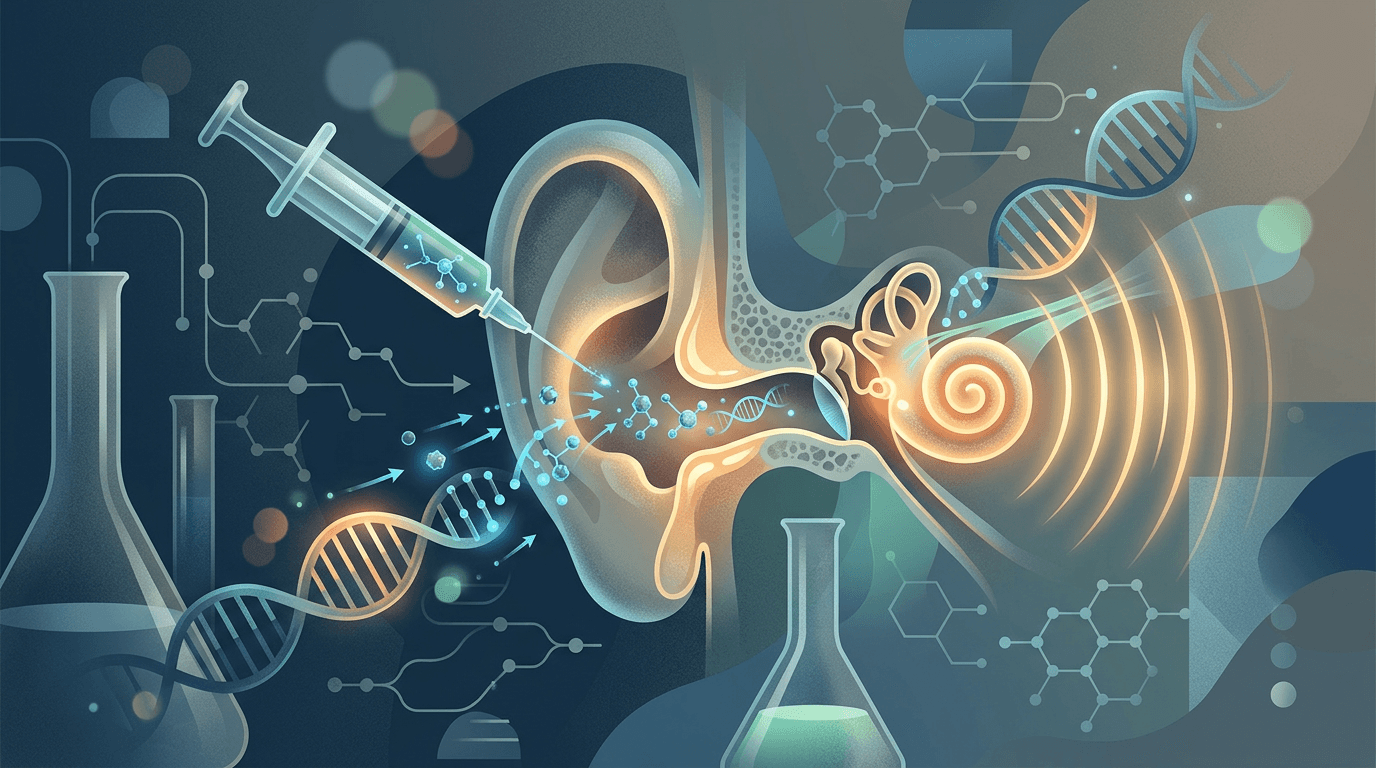
On April 24, Regeneron scored a gene therapy approval from the FDA. The drug, called Otarmeni (good luck pronouncing the generic name: lunsotogene parvec-cwha), treats severe-to-profound hearing loss caused by mutations in both copies of the OTOF gene.
This gene normally produces a protein called otoferlin, which acts like a tiny relay switch inside your inner ear. Without it, sound waves hit the ear just fine, but the signal never gets forwarded to the brain. Think of it like having a perfectly good antenna on your roof with a severed cable to the TV. The broadcast is there; the connection isn't.
Otarmeni fixes the cable. A surgeon delivers the therapy directly into the cochlea (the snail-shaped structure in your inner ear) through a one-time infusion. The treatment carries a working copy of the OTOF gene, packaged inside a harmless viral vector called AAV, which teaches inner hair cells to start producing otoferlin again.
One surgery. That's it.
The Numbers That Got the FDA Moving
The approval was based on a single-arm trial of 24 pediatric patients, ranging from 10 months to 16 years old. Of the 20 patients who could be evaluated, 70% met the key secondary ABR endpoint. Some of these kids went from not hearing anything below a shout to picking up conversational speech.
Before Otarmeni, the only options were hearing aids (which often don't work well for this condition, since the problem isn't volume but signal transmission) and cochlear implants. Neither one actually fixes the underlying cause. They're workarounds, not solutions.
The FDA clearly agreed this was significant. Otarmeni received a full stack of regulatory designations: orphan drug, rare pediatric disease, fast track, and RMAT (regenerative medicine advanced therapy). The agency approved it just , tying for the fastest modern BLA review on record. That's the FDA equivalent of skipping the line at a nightclub.
More from this issue
View full issue →
Clinical & Regulatory5 min read
Regeneron Was the Last Holdout. The White House Just Got Its Deal.
Regeneron was the 17th and final major drugmaker to cut a pricing deal with the White House's Most Favored Nation initiative. The agreement includes a 58% price cut on Praluent, a free gene therapy for rare childhood deafness, and nearly $10 billion in domestic manufacturing pledges. With 86% of the branded drug market now covered, the real question is what these deals actually mean for patients at the pharmacy counter.

Get tomorrow's biotech intelligence before your competitors.
Join thousands of biotech professionals who start their day with our free, daily briefing.
