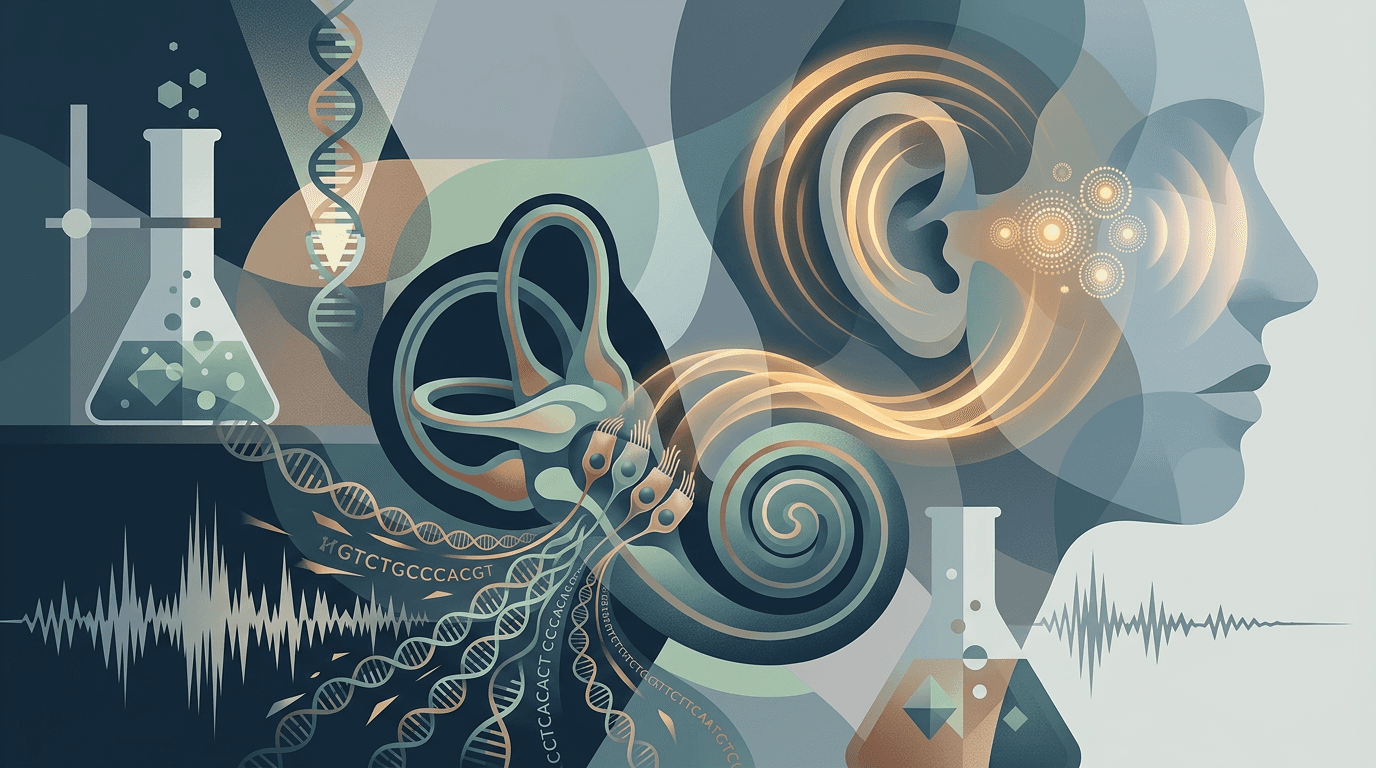
The Gene Therapy That's Making Deaf People Hear Whispers
A gene therapy trial just restored hearing in 90% of deaf patients, some within weeks of a single injection. The results are 'mind-boggling,' and pharma companies are spending billions to win the race.
Imagine spending your entire life in silence. No music, no conversation, no birdsong. Then someone injects a tiny virus into your inner ear, and within weeks, you can hear a whisper.
That's not science fiction. It just happened to roughly 90% of patients in the largest gene therapy trial ever conducted for inherited deafness. The results, published April 22, 2026, in Nature, are being called "mind-boggling" by the researchers involved. And they might signal the beginning of the end for one of medicine's most stubbornly untreatable conditions.
From Profound Silence to Casual Conversation
The trial enrolled 42 patients across eight sites in China, ranging in age from less than a year old to 32. Every single one of them had DFNB9, a form of genetic deafness caused by mutations in the OTOF gene. That gene is supposed to produce a protein called otoferlin, which acts like a relay switch between your inner ear and your brain. Without it, sound waves hit the ear just fine, but the signal never reaches the brain. Think of it like having a perfectly good TV with a broken HDMI cable.
Each patient received a single injection of a virus carrying a working copy of the OTOF gene directly into the inner ear. The virus (an AAV, or adeno-associated virus) is basically a biological FedEx truck: it delivers the genetic payload, the cells start making otoferlin, and the relay switch flicks on.
The results were striking. Before treatment, the average hearing threshold was greater than 95 decibels, which is essentially profound deafness. After treatment, patients achieved an average improvement of roughly 52 decibels, with post-treatment thresholds ranging from 30 to 58 decibels, squarely in the range of normal conversation. Some patients could even hear whispers. And these gains held steady for up to 2.5 years.
Kids Responded Best (But Adults Surprised Everyone)
Younger children with healthier inner ears saw the biggest improvements, which makes intuitive sense. Their brains are still wiring up their auditory circuits, so restoring the signal early gives the brain more to work with. Several children developed speech skills for the first time. An 11-year-old gained rudimentary speech after a lifetime without it.
More from this issue
View full issue →
Deals & M&A6 min read
Lilly's $300M Bet on a Biotech That Doesn't Even Have a Drug in Humans Yet
Eli Lilly just paid up to $300 million for a three-year-old startup that hasn't tested a single drug in humans. CrossBridge Bio's dual-payload ADC technology is so hot that Lilly couldn't wait for clinical data, and the deal reveals just how frenzied the ADC arms race has become.


Get tomorrow's biotech intelligence before your competitors.
Join thousands of biotech professionals who start their day with our free, daily briefing.