J&J Found a Way to Make Your Immune System Take Out Its Own Trash
J&J's nipocalimab just scored its fifth FDA Fast Track designation, this time for lupus. It works by blocking the receptor that keeps harmful autoantibodies alive, and the Phase 2 data suggest it could finally help patients escape the steroid trap that defines current treatment.
Lupus is the autoimmune disease that Hollywood loves to joke about ("it's never lupus") but patients absolutely do not find funny. About 3 to 5 million people worldwide live with systemic lupus erythematosus (SLE), and achieving the holy grail of treatment — lasting remission without steroids — remains elusive for most. Johnson & Johnson just got a green light to try to change that.
On March 3, the FDA granted Fast Track designation to nipocalimab for treating adults with SLE. That's the agency's way of saying: "This looks promising, and the need is urgent. Let's speed things up." It's also the fifth Fast Track designation nipocalimab has earned across different diseases, which is starting to feel less like a drug and more like a Swiss Army knife.
The Antibody Recycling Problem
To understand why nipocalimab matters, you need to understand one weird quirk of human biology.
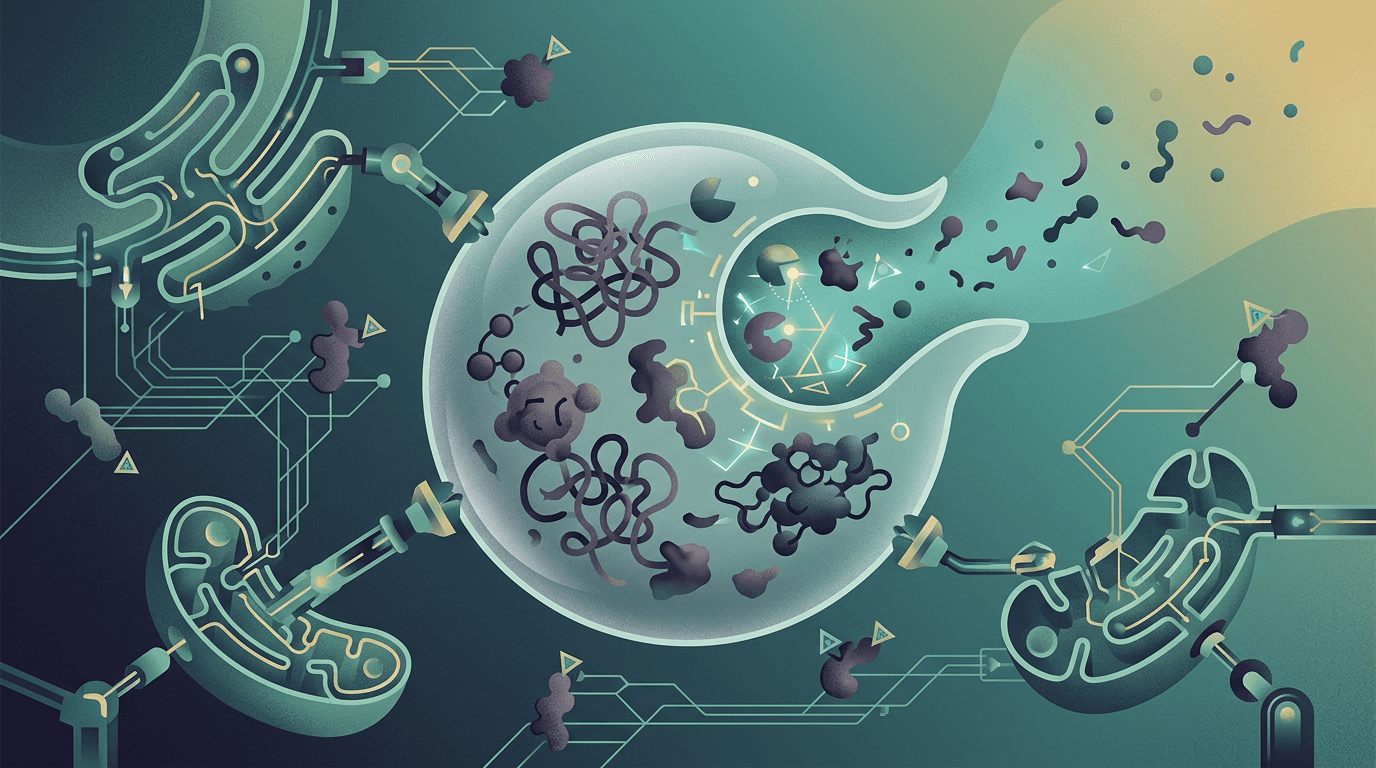
Your body has a receptor called FcRn (neonatal Fc receptor, if you want the full name) that acts like a bouncer at a nightclub. Its job is to grab IgG antibodies, the workhorses of your immune system, and recycle them back into your bloodstream instead of letting them get destroyed. Normally, this is great. Antibodies last longer, and your immune system stays strong.
But in lupus, the system goes haywire. Your body produces autoantibodies, rogue IgG molecules that attack your own tissues: kidneys, skin, joints, brain. And FcRn dutifully recycles those bad actors right along with the good ones. It's like a recycling plant that can't tell the difference between clean water and toxic waste; it just keeps pumping everything back into the supply.
Nipocalimab blocks FcRn. When FcRn can't do its recycling job, IgG antibodies (including the harmful autoantibodies) get broken down instead of preserved. Think of it as cutting the power to that recycling plant. The toxic waste finally gets disposed of.
What makes this clever is the selectivity. Nipocalimab was engineered to avoid triggering broader immune reactions, so it doesn't carpet-bomb your entire defense system the way some immunosuppressants do. It just stops the recycling.
More from this issue
View full issue →
Clinical & Regulatory4 min read
The FDA Just Made It Way Cheaper to Copy Blockbuster Drugs
The FDA just proposed letting biosimilar developers skip expensive U.S.-only comparison studies, potentially saving $20 million per program. Combined with other recent reforms, this could cut total development costs in half and reshape competition for blockbuster biologic drugs.


Get tomorrow's biotech intelligence before your competitors.
Join thousands of biotech professionals who start their day with our free, daily briefing.