The Drug That Learned to Pick a Lock No One Could Open
For 20 years, kids with Hunter syndrome had a treatment that could help their bodies but couldn't reach their brains. Denali Therapeutics just got FDA approval for the first biologic designed to cross the blood-brain barrier, and the implications go way beyond one rare disease.
For nearly two decades, kids with Hunter syndrome had exactly one FDA-approved treatment. It could help their livers. It could help their lungs. But their brains? Off limits.
That's because the blood-brain barrier, the body's ultra-selective bouncer for the central nervous system, doesn't let big therapeutic molecules through. Enzymes that could fix the rest of the body got turned away at the door. Meanwhile, the neurological damage kept progressing.
On March 25, 2026, that changed. The FDA granted accelerated approval to Avlayah (tividenofusp alfa-eknm), a new enzyme replacement therapy from Denali Therapeutics. It's the first approved biologic specifically designed to cross the blood-brain barrier. Think of it as a delivery truck that finally figured out the password to get past the gate.
The Disease Nobody Talks About
Hunter syndrome, also called MPS II, is a rare genetic disorder that affects roughly 1 in 100,000 to 170,000 male births. The disease is caused by a missing or broken enzyme called iduronate 2-sulfatase (IDS), which the body needs to break down complex sugars. Without it, those sugars pile up in cells throughout the body like trash in a house with no garbage pickup.
The existing treatment, idursulfase (brand name Elaprase), has been around since 2006. It replaces the missing enzyme through an IV infusion. The problem: the enzyme can't cross the blood-brain barrier. For kids with the neuronopathic form of the disease, cognitive decline, behavioral changes, and neurological deterioration continue unchecked. Parents watch their children slip away, knowing the available treatment simply can't reach the part of the body that matters most.
Avlayah is the first new Hunter syndrome therapy in almost 20 years. And it was built to solve exactly this problem.
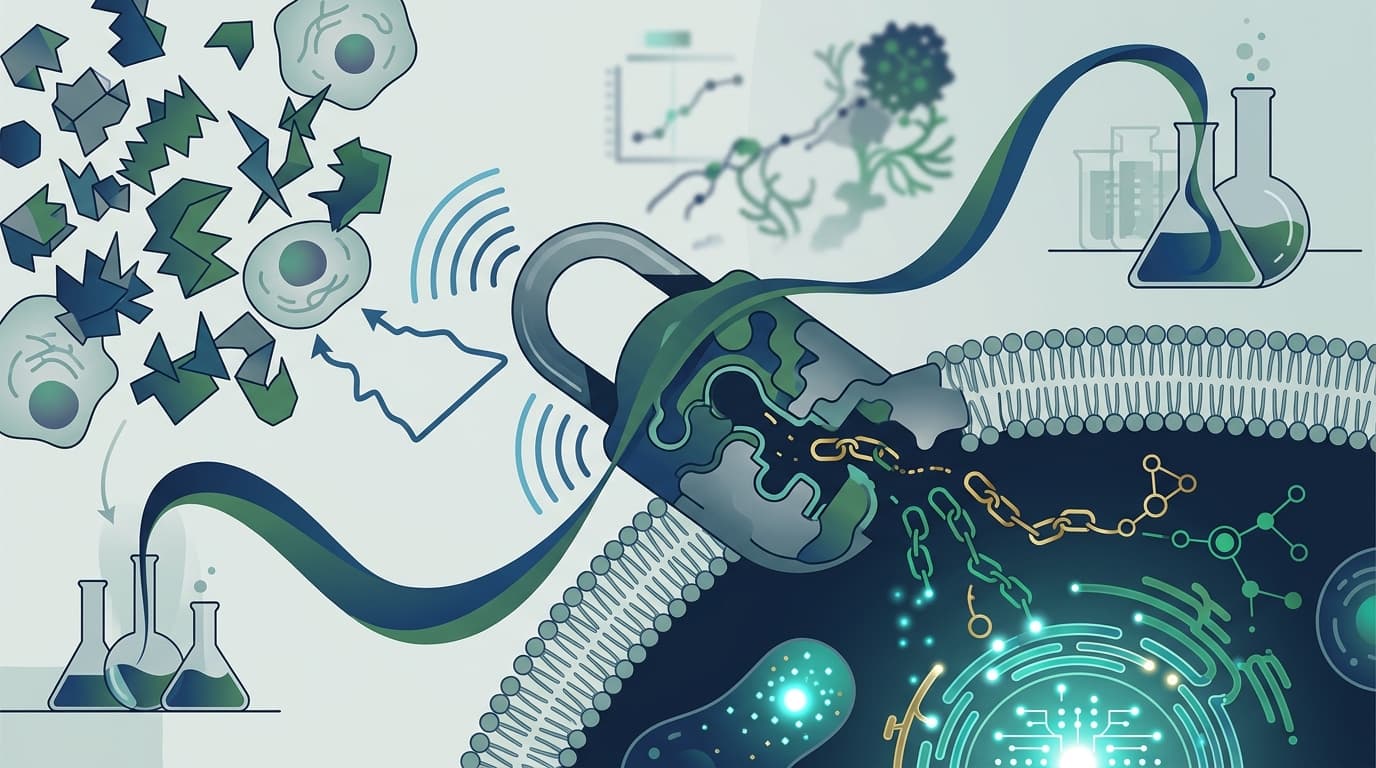
How You Sneak Past the Brain's Bouncer
Denali's secret weapon is its Transport Vehicle (TV) platform, a clever piece of protein engineering that essentially hitchhikes into the brain on an existing delivery route.
Your brain needs iron to function. To get it, iron-carrying proteins called transferrin bind to transferrin receptors on the surface of the blood-brain barrier's endothelial cells. Those cells pull the iron across and into the brain. It's like a VIP entrance that only opens for specific guests.
More from this issue
View full issue →
Funding & Financings4 min read
The Billion-Dollar Startup That Wants to Dethrone Wegovy
Kailera Therapeutics raised nearly $1 billion in private funding before most people knew its name. Now the obesity-focused biotech is going public, and its IPO could reveal whether Wall Street's love affair with weight-loss drugs still has room to grow.

Clinical & Regulatory

Get tomorrow's biotech intelligence before your competitors.
Join thousands of biotech professionals who start their day with our free, daily briefing.