
The Tiny Biotech That Made Colorectal Cancer Flinch
A masked antibody-drug conjugate from tiny CytomX Therapeutics just posted a 32% response rate in late-line colorectal cancer, a disease where low-single-digit responses are the norm. The stock went haywire, but the science behind it might be even more interesting.
A Stock Chart That Tells a Story
CytomX Therapeutics was a $4.68 stock. The kind of small biotech that most investors scroll past without a second thought. Then, on March 16, the company dropped Phase 1 data for its colorectal cancer drug, and the stock ripped 65% intraday before settling at a 44% gain.
The days that followed were chaotic, with sharp drops and bounces throughout the week. Classic biotech volatility. But behind the trading noise, something genuinely interesting happened. A tiny company with a clever idea just posted results in one of oncology's toughest neighborhoods, and the numbers were good enough to make Wall Street pay attention.
Why Late-Line Colorectal Cancer Is a Desert
To appreciate what CytomX pulled off, you need to understand the playing field. Late-line metastatic colorectal cancer (CRC) is where patients end up after burning through two or three rounds of chemo. The options are grim.
The current go-to combo, trifluridine/tipiracil plus bevacizumab, delivers a median overall survival of about 10.8 months. Fruquintinib, another option, manages 7.4 months. And for patients without specific genetic markers, response rates hover in the low single digits. That means the vast majority of patients take these drugs and their tumors don't shrink at all.
So when a new drug walks into that desert and starts producing meaningful responses, people notice.
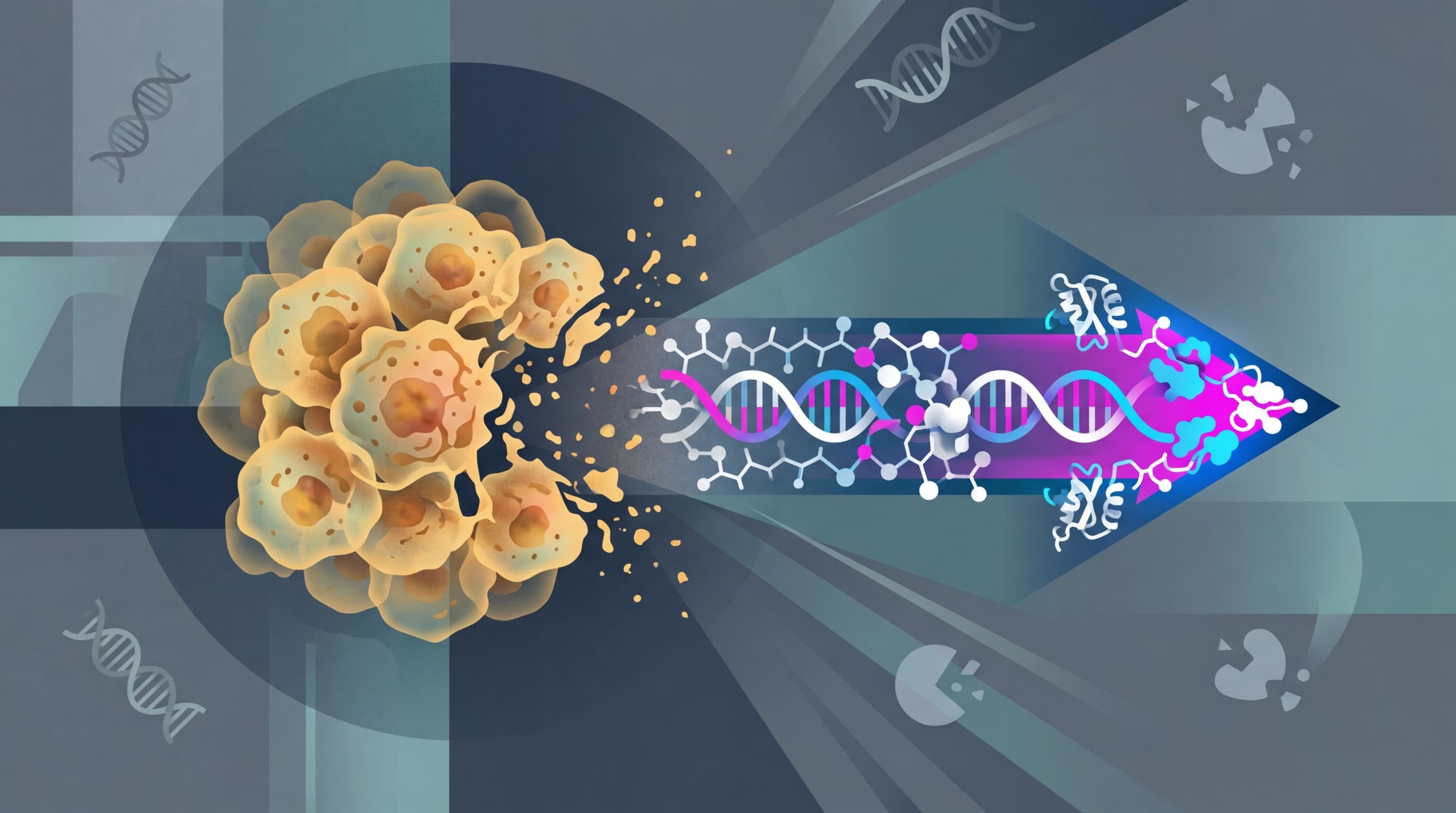
The Drug With a Disguise
CytomX's candidate is called Varseta-M (varsetatug masetecan), and it's built on an idea that sounds almost too simple to work. It's an antibody-drug conjugate, or ADC: basically a guided missile that attaches a toxic payload to an antibody designed to find cancer cells. ADCs are one of the hottest drug categories in oncology right now, with the market projected to hit $20-30 billion by the early 2030s.
But ADCs have a persistent problem. The targets they chase (proteins on cancer cells) often show up on healthy cells too. That means the toxic payload can damage normal tissue, causing nasty side effects. It's like hiring a hitman who's great at finding the target but also shoots bystanders.
More from this issue
View full issue →
Clinical & Regulatory4 min read
The $3B Startup That Wants to Dethrone Robotic Surgery's King
CMR Surgical just launched its Versius Plus robotic system in the U.S., taking direct aim at Intuitive Surgical's $10 billion empire. With modular arms that roll between operating rooms and $1.4 billion in funding, this UK challenger is betting that hospitals are ready for a more flexible (and potentially cheaper) alternative.


Get tomorrow's biotech intelligence before your competitors.
Join thousands of biotech professionals who start their day with our free, daily briefing.
