The Cancer Drug That Plays Dead Until It Finds Its Target
Astellas just dropped $335 million upfront on a cancer drug that stays completely dormant until it reaches the tumor; then it wakes up and attacks. The early data on VIR-5500 is turning heads, and the deal's timing tells you everything about Astellas' post-XTANDI anxiety.
T-cell engagers have a dirty secret: they're great at killing cancer, and terrible at leaving everything else alone.
These drugs work by grabbing a T cell (your immune system's hitman) with one arm and a cancer cell with the other, forcing an introduction that ends badly for the tumor. The problem is first-generation versions can't tell the difference between cancer cells and healthy tissue that happens to express the same protein. It's like hiring a bouncer who throws out everyone wearing a red shirt, not just the troublemaker.
Vir Biotechnology thinks it's solved that problem, and Astellas just wrote a check that suggests they agree.
A $1.7 Billion Bet on a Disguise
Astellas is paying $335 million upfront for the rights to VIR-5500, a masked T-cell engager targeting prostate cancer. The total deal could reach $1.7 billion with milestones, plus tiered double-digit royalties on sales outside the U.S.
That upfront number breaks down in an interesting way: $240 million in cash, $75 million as an equity investment in Vir at a 50% premium to market price, and a $20 million near-term milestone. When a pharma company buys your stock at a 50% markup, that's not just a licensing deal — that's a statement of conviction.
The two companies will split U.S. profits 50/50, with Astellas leading commercialization. Outside the U.S., Astellas gets exclusive rights. Development costs get shared 60/40, with Astellas picking up the bigger tab. Vir keeps running the ongoing Phase 1 trial until it hands the baton to Astellas, who then quarterbacks everything from there.
The Cloaking Device That Makes This Drug Different
So what exactly is Astellas paying for?
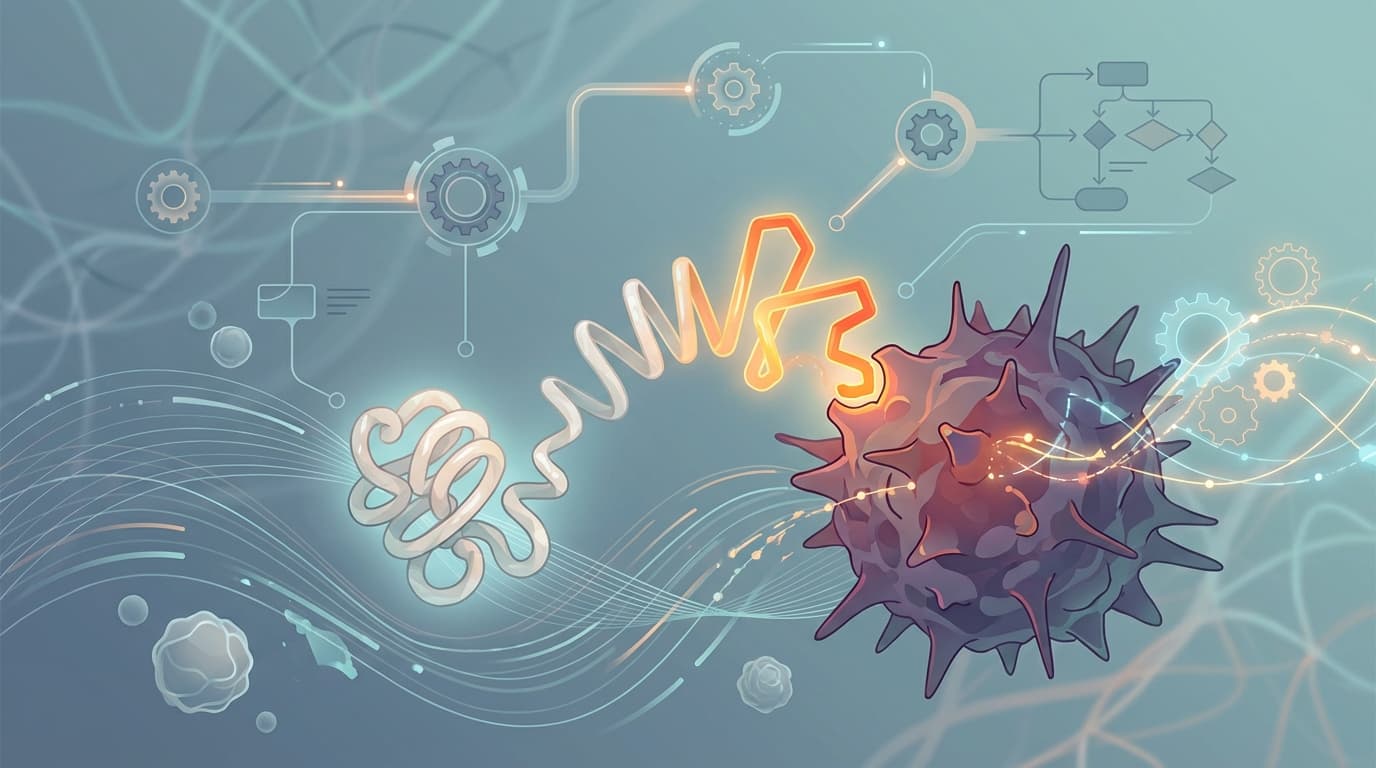
VIR-5500 uses something called PRO-XTEN masking technology, essentially a molecular disguise. Think of it like a grenade with the pin permanently locked in place. The drug circulates through your body in a "masked" state, completely inert. It can't grab T cells. It can't grab anything. It's playing dead.
But when VIR-5500 reaches the tumor, enzymes that tumors naturally produce (called proteases) chew through the mask. The drug wakes up, locks onto PSMA, a protein covering prostate cancer cells, and starts recruiting T cells to attack.
More from this issue
View full issue →
Neurology and Digital Health5 min read
The $130M Bet on the Migraine Patients That Nobody Can Help
Half of migraine patients don't respond to CGRP drugs, the only biologic game in town. A brand-new startup just launched with $130 million to target a completely different brain pathway, and it could change the treatment map entirely.

Regulatory and Policy

Get tomorrow's biotech intelligence before your competitors.
Join thousands of biotech professionals who start their day with our free, daily briefing.